- Metallic: First Generation: Urospiral (Fabian,1980). Second: Prosta-Coil (Self-expanding). Memo-kath (Thermal expanding). Third Generation: Memo-kath 028 (Memory)
- Plastic Stents: The Spanner (Temporary)
- Biodegradable Stents: Poly-glycolic Acid: By time degraded to CO2 and water. No need for removal.
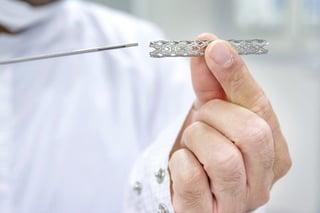
Prostatic Stents Contra-indications:
Relative contraindications to the procedure include:
- Meatal and urethral strictures
- Active urinary tract infection (UTI)
- Haematuria and patients on anti-coagulants that prevents adequate visualisation for placement
- Bladder calculi
- A large Prostatic median lobe that hampers satisfactory placement
- Bladder neck stenosis
- A prostatic urethra shorter than 2cm (Fibrous prostate) as the least product length is 3 Cm.
Complications of Prostatic Stents:
- As with any surgical procedure there are potential complications, which can include: perineal discomfort, light bleeding, post-void dribbling. These complications are common and usually subside after few days.
- More serious complications can include: Stent migration into the distal urethra or bladder, encrustation and stone formation on the stent that can obstruct the lumen.
Indications and exclusion criteria:
- Included severe respiratory and cardiovascular disease.
- Excluded bladder carcinoma and prostatic carcinoma, calculi or detrusor failure
- Normal coagulation profile (Aspirin and Plavix are to be stopped for a week prior to the procedure)
- Warfarin has to be replaced with low molecular wt. Heparin and stopped 12 hours prior to the procedure
Conclusion:
The Memo-kath intra-prostatic stent is a valuable addition to the armamentarium of the urologist treating elderly men with advanced bladder outlet obstruction and associating major contra-indications for general and regional anaesthesia.
Learn more about our Editorial Process.
Topic: Urology Blogs









Leave a comment